Ernest Rutherford was born on August 30, 1871, in Nelson, New Zealand. 1894, he was awarded an 1851 Exhibition Science Scholarship, enabling him to go to Trinity College, Cambridge, as a research student at the Cavendish Laboratory under J.J. Thomson.[1]
Rutherford's Experiment
At the time of Rutherford’s ‘Gold Foil Experiment’ he knew that radioactive substances naturally emitted radiation that could pass through thin layers of materials but not thicker layers. This radiation contained alpha particles (α). Atoms which have lost their electrons making them positively charged. At Manchester, Rutherford worked in collaboration with colleague in his department, German physicist Hans Geiger. Together they invented a method to detect a single α particle and for counting the number α particles emitted by radioactive source.[2]
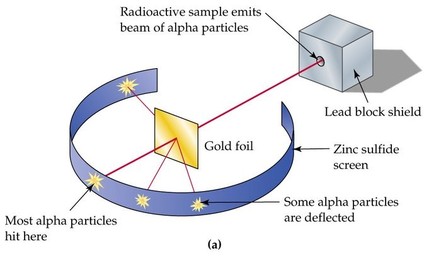
Rutherford and Geiger fired a narrow beam of high speed α particles into a fine tissue like sheet of Gold. The α particles were detected on a moveable curved screen that was coated in zinc sulphide. The substance is fluorescent and gives a tiny flash of light (called a scintillation) when hit by an electron.[3] At first they placed a screen behind the gold foil. Rutherford reasoned that if the positively charged material in each gold atom is spread out over the entire volume of the atom, as J.J Thomas had proposed, its electric field would not be intense enough to repel the tiny, very fast moving, positively charged α particle to any significant degree. Instead they would pass straight through the larger gold atoms.[4]
At the time of Rutherford’s ‘Gold Foil Experiment’ he knew that radioactive substances naturally emitted radiation that could pass through thin layers of materials but not thicker layers. This radiation contained alpha particles (α). Atoms which have lost their electrons making them positively charged. At Manchester, Rutherford worked in collaboration with colleague in his department, German physicist Hans Geiger. Together they invented a method to detect a single α particle and for counting the number α particles emitted by radioactive source.[2]
Rutherford and Geiger fired a narrow beam of high speed α particles into a fine tissue like sheet of Gold. The α particles were detected on a moveable curved screen that was coated in zinc sulphide. The substance is fluorescent and gives a tiny flash of light (called a scintillation) when hit by an electron.[3] At first they placed a screen behind the gold foil. Rutherford reasoned that if the positively charged material in each gold atom is spread out over the entire volume of the atom, as J.J Thomas had proposed, its electric field would not be intense enough to repel the tiny, very fast moving, positively charged α particle to any significant degree. Instead they would pass straight through the larger gold atoms.[4]
First Insight into the Nucleus
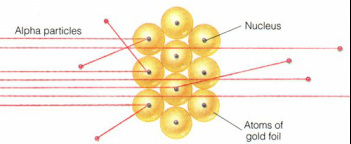
After analysing the data, Rutherford concluded that the results of the α particle scattering experiment could be explained only if the positively charged atom was not spread over its whole volume but concentrated in a small volume at the centre. He called concentrated region of positively charged the nucleus. He then proposed that most of an atom is empty space, so when alpha particle moves through this empty space the nucleus has a very little effect on where it goes. The α particle will only move significantly if it gets close to the nucleus.[5]
After analysing the data, Rutherford concluded that the results of the α particle scattering experiment could be explained only if the positively charged atom was not spread over its whole volume but concentrated in a small volume at the centre. He called concentrated region of positively charged the nucleus. He then proposed that most of an atom is empty space, so when alpha particle moves through this empty space the nucleus has a very little effect on where it goes. The α particle will only move significantly if it gets close to the nucleus.[5]
Rutherford's Atomic Model
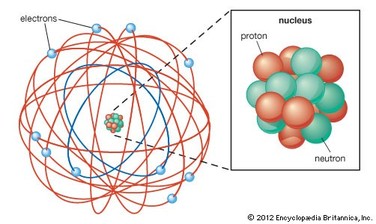
From this Rutherford realised that the plum pudding model could not account for this discrepancy. In 1911 proposed a new and improved atomic model. In this model, the negative electrons orbited around a tiny, centralized, positively charged and incredibly dense and small central called the nucleus. This was the first of what can be referred to as “solar system” models, where the electrons orbit the nucleus like the planets orbit the sun. [6]
From this Rutherford realised that the plum pudding model could not account for this discrepancy. In 1911 proposed a new and improved atomic model. In this model, the negative electrons orbited around a tiny, centralized, positively charged and incredibly dense and small central called the nucleus. This was the first of what can be referred to as “solar system” models, where the electrons orbit the nucleus like the planets orbit the sun. [6]
Other Discoveries
Rutherford also designed a series of experiments to determine the size of the nucleus compared with that of an atom. He measured the angles of scattering and their frequency by using slits of various foils and different metals. He used mathematical modelling to deduce the positive charge had to be concentrated in a sphere less than 10-14m in diameter.[7] Rutherford had also heavily contributed to the Field of Physics. He successfully marked out a wholly new branch of physics called radioactivity with Professor Thomson working together they studied the behaviour and mobility of ions in gases. During World War I Rutherford also turned to solving problems connected with submarine detection.[8]
Rutherford also designed a series of experiments to determine the size of the nucleus compared with that of an atom. He measured the angles of scattering and their frequency by using slits of various foils and different metals. He used mathematical modelling to deduce the positive charge had to be concentrated in a sphere less than 10-14m in diameter.[7] Rutherford had also heavily contributed to the Field of Physics. He successfully marked out a wholly new branch of physics called radioactivity with Professor Thomson working together they studied the behaviour and mobility of ions in gases. During World War I Rutherford also turned to solving problems connected with submarine detection.[8]
[1] Media, N. 2014. Ernest Rutherford - Biographical. [online] Available at: http://www.nobelprize.org/nobel_prizes/chemistry/laureates/1908/rutherford-bio.html [Accessed: 10 Mar 2014].
[2] Sharwood, J. and Corrigan, D. 2007. Nelson chemistry. South Melbourne: Thomson Learning.
[3] Aip.org. 2014. Rutherford's Nuclear World: The Story of the Discovery of the Nucleus | Sections | American Institute of Physics. [online] Available at: http://www.aip.org/history/exhibits/rutherford/sections/alpha-particles-atom.html [Accessed: 10 Mar 2014].
[4] P.Kilman. 2014. [online] Available at: http://undsci.berkeley.edu/lessons/pdfs/rutherford.pdf [Accessed: 10 Mar 2014].
[5] Sharwood, J. and Corrigan, D. 2007. Nelson chemistry. South Melbourne: Thomson Learning.
[6] P.Kilman. 2014. [online] Available at: http://undsci.berkeley.edu/lessons/pdfs/rutherford.pdf [Accessed: 10 Mar 2014].
[7] NZEDGE.COM. 2014. Ernest Rutherford. [online] Available at: http://www.nzedge.com/ernest-rutherford/ [Accessed: 10 Mar 2014].
[8] Chalmers, A. 2014. The Rutherford Journal - The New Zealand Journal for the History and Philosophy of Science and Technology. [online] Available at: http://www.rutherfordjournal.org/article010101.html [Accessed: 11 Mar 2014].
[2] Sharwood, J. and Corrigan, D. 2007. Nelson chemistry. South Melbourne: Thomson Learning.
[3] Aip.org. 2014. Rutherford's Nuclear World: The Story of the Discovery of the Nucleus | Sections | American Institute of Physics. [online] Available at: http://www.aip.org/history/exhibits/rutherford/sections/alpha-particles-atom.html [Accessed: 10 Mar 2014].
[4] P.Kilman. 2014. [online] Available at: http://undsci.berkeley.edu/lessons/pdfs/rutherford.pdf [Accessed: 10 Mar 2014].
[5] Sharwood, J. and Corrigan, D. 2007. Nelson chemistry. South Melbourne: Thomson Learning.
[6] P.Kilman. 2014. [online] Available at: http://undsci.berkeley.edu/lessons/pdfs/rutherford.pdf [Accessed: 10 Mar 2014].
[7] NZEDGE.COM. 2014. Ernest Rutherford. [online] Available at: http://www.nzedge.com/ernest-rutherford/ [Accessed: 10 Mar 2014].
[8] Chalmers, A. 2014. The Rutherford Journal - The New Zealand Journal for the History and Philosophy of Science and Technology. [online] Available at: http://www.rutherfordjournal.org/article010101.html [Accessed: 11 Mar 2014].