James Chadwick was born in Cheshire, England, on 20th October. He graduated from the Honours School of Physics in 1911 and spent the next two years under Professor Ernest Rutherford in the Physical Laboratory in Manchester.[1]
Chadwick's Predecessors
Chadwick had an abundance of knowledge he had gained from previous discoveries, which help him discover the neutron. With Ernest Rutherford’s discovery of the positively-charged proton, as well as J.J Thompson’s discovery of the electron, it seemed that the atomic theory structure was almost completed. With just these basic elements of the atomic structure, it was clear to him why atoms appeared to be neutral as the negative charge of the electron and the positive charge of the proton cancelled out. In 1920, the possibility of the existence of a neutrally-charged particle with a similar mass to that of a proton. This would help to keep the atom neutral, and to fix some differences found between the atomic number of an atom and its atomic mass.
Chadwick had an abundance of knowledge he had gained from previous discoveries, which help him discover the neutron. With Ernest Rutherford’s discovery of the positively-charged proton, as well as J.J Thompson’s discovery of the electron, it seemed that the atomic theory structure was almost completed. With just these basic elements of the atomic structure, it was clear to him why atoms appeared to be neutral as the negative charge of the electron and the positive charge of the proton cancelled out. In 1920, the possibility of the existence of a neutrally-charged particle with a similar mass to that of a proton. This would help to keep the atom neutral, and to fix some differences found between the atomic number of an atom and its atomic mass.
Chadwick's Experiment
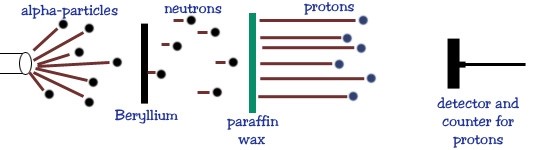
In 1932, Chadwick carried out an experiment in which a sample of Beryllium was bombarded with alpha particles, which caused it to emit this mysterious radiation. He assumed it was gamma rays.[2] He then discovered that this radiation, upon striking Paraffin Wax (proton-rich surface), would dislodged some of the protons, coming to the conclusion it was gamma rays. Chadwick, however did not believe that gamma rays could account for the protons from the wax. He was convinced that the beryllium was emitting neutrons.[3]
Chadwick could tell that the mysterious radiation was neutral because it was not deflected by electric or magnetic fields and it did not invoke the photoelectric effect, unlike standard gamma radiation. This was when photons such as gamma rays, strike certain surfaces, they discharge electrons.[4] However it discharged protons, which meant that the particles had to be more massive than previously expected. Therefore also coming to the conclusion that neutrons had a heavier mass than protons.
In 1932, Chadwick carried out an experiment in which a sample of Beryllium was bombarded with alpha particles, which caused it to emit this mysterious radiation. He assumed it was gamma rays.[2] He then discovered that this radiation, upon striking Paraffin Wax (proton-rich surface), would dislodged some of the protons, coming to the conclusion it was gamma rays. Chadwick, however did not believe that gamma rays could account for the protons from the wax. He was convinced that the beryllium was emitting neutrons.[3]
Chadwick could tell that the mysterious radiation was neutral because it was not deflected by electric or magnetic fields and it did not invoke the photoelectric effect, unlike standard gamma radiation. This was when photons such as gamma rays, strike certain surfaces, they discharge electrons.[4] However it discharged protons, which meant that the particles had to be more massive than previously expected. Therefore also coming to the conclusion that neutrons had a heavier mass than protons.
Chadwick's Atomic Model
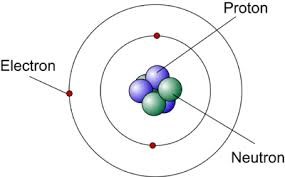
Neutrons were found to have the same mass as protons which accounted for more of the mass of the atom and allowed the known mass of an atom and the known mass of its particles to match. The common understanding of an atom was now a nucleus containing positively charged protons and neutral neutrons with the rest of the atom being space in which negatively charged electrons, with a mass so small compared to that of the nucleus "orbit" the nucleus on energy levels corresponding to the amount of energy the electrons hold.[5]
Neutrons were found to have the same mass as protons which accounted for more of the mass of the atom and allowed the known mass of an atom and the known mass of its particles to match. The common understanding of an atom was now a nucleus containing positively charged protons and neutral neutrons with the rest of the atom being space in which negatively charged electrons, with a mass so small compared to that of the nucleus "orbit" the nucleus on energy levels corresponding to the amount of energy the electrons hold.[5]
Other Discoveries
In 1932, Chadwick made a fundamental discovery in the domain on nuclear science. Neutrons were involved in creating nuclear explosions and nuclear energy, for it is by bombardment with high-energy neutrons that scientists first learned how to split an atom. Chadwick in this way prepared the way towards the fission of uranium 235 and towards the creation of the atomic bomb.[6]
In 1932, Chadwick made a fundamental discovery in the domain on nuclear science. Neutrons were involved in creating nuclear explosions and nuclear energy, for it is by bombardment with high-energy neutrons that scientists first learned how to split an atom. Chadwick in this way prepared the way towards the fission of uranium 235 and towards the creation of the atomic bomb.[6]
[1] Nobelprize.org. 2014. James Chadwick - Biographical. [online] Available at: http://www.nobelprize.org/nobel_prizes/physics/laureates/1935/chadwick-bio.html [Accessed: 10 Mar 2014].
[2] Mcphee, I. 2014. The Discovery of the Neutron. [online] Available at: https://suite101.com/a/the-discovery-of-the-neutron-a46060 [Accessed: 10 Mar 2014].
[3] Www-outreach.phy.cam.ac.uk. 2014. Cambridge Physics - Discovery of the Neutron. [online] Available at: http://www-outreach.phy.cam.ac.uk/camphy/neutron/neutron4_1.htm [Accessed: 10 Mar 2014].
[4] Colwell, C. 2014. PhysicsLAB: Famous Experiments: The Discovery of the Neutron. [online] Available at: http://dev.physicslab.org/Document.aspx?doc&filename=AtomicNuclear_ChadwickNeutron.xml [Accessed: 10 Mar 2014].
[5] Harrison,, A. 2012. NEUTRON SCIENCE. New Scientist, 216 (2893), pp. 23-38.
[6] Brown, A. and Stuewer, R. R. 2008. The neutron and the bomb: a biography of Sir James Chadwick. Physics Today, 50 (12), pp. 65--66.
[2] Mcphee, I. 2014. The Discovery of the Neutron. [online] Available at: https://suite101.com/a/the-discovery-of-the-neutron-a46060 [Accessed: 10 Mar 2014].
[3] Www-outreach.phy.cam.ac.uk. 2014. Cambridge Physics - Discovery of the Neutron. [online] Available at: http://www-outreach.phy.cam.ac.uk/camphy/neutron/neutron4_1.htm [Accessed: 10 Mar 2014].
[4] Colwell, C. 2014. PhysicsLAB: Famous Experiments: The Discovery of the Neutron. [online] Available at: http://dev.physicslab.org/Document.aspx?doc&filename=AtomicNuclear_ChadwickNeutron.xml [Accessed: 10 Mar 2014].
[5] Harrison,, A. 2012. NEUTRON SCIENCE. New Scientist, 216 (2893), pp. 23-38.
[6] Brown, A. and Stuewer, R. R. 2008. The neutron and the bomb: a biography of Sir James Chadwick. Physics Today, 50 (12), pp. 65--66.