Joseph John Thomson a British physicist was born in Cheetham Hill, Manchester on December 18, 1856. He was a man who had enrolled in the prestigious school of Cambridge and he remained a member of the College for the rest of his life, becoming Lecturer of experimental physics.[1]
Cathode Rays
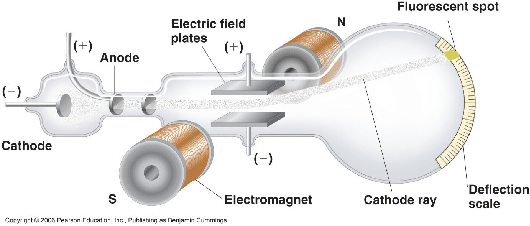
Thomson was investigating with a long-standing puzzle known as "cathode rays" found because of British scientist Michael Faraday in 1838. These were currents of electricity inside empty glass tubes that had most of its air pumped out, leaving a strange light to be produced between two electrodes. These two electrodes were known as the Cathode (negatively charged electrodes and Anodes (positively charged electrodes). [2]
Thomson positioned electrodes and a magnet around a discharge tube to investigate the charge and mass of the particles that make up the cathode rays. Thomson did this by measuring how much the particles in the cathode rays were deflected by different electrical and magnetic fields.[3] He was then able to bend the ray using a magnetic field and then measure the direction the ray bent so that he could determine both how much mass was involved and how it was charged.
Thomson was investigating with a long-standing puzzle known as "cathode rays" found because of British scientist Michael Faraday in 1838. These were currents of electricity inside empty glass tubes that had most of its air pumped out, leaving a strange light to be produced between two electrodes. These two electrodes were known as the Cathode (negatively charged electrodes and Anodes (positively charged electrodes). [2]
Thomson positioned electrodes and a magnet around a discharge tube to investigate the charge and mass of the particles that make up the cathode rays. Thomson did this by measuring how much the particles in the cathode rays were deflected by different electrical and magnetic fields.[3] He was then able to bend the ray using a magnetic field and then measure the direction the ray bent so that he could determine both how much mass was involved and how it was charged.
Thompson's Discovery and Atomic Model
From this he discovered that no matter what metal the electrodes were made up of or what type of gas was in the tube, the mass to charge ratio was unchanged. He also deducted that there were two parts of atom held together by electrostatic attraction. A heavier part, responsible for the majority of mass of the atom and a negatively charged ‘corpuscle’ (electron).[4] This meant that Dalton’s model of an atom was incorrect, since the atom was not the smallest possible particle.
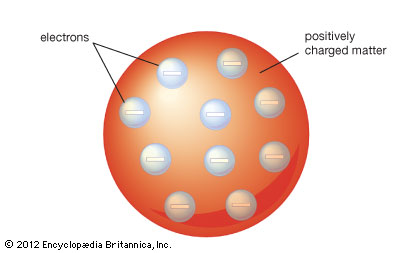
His experiment findings drove Thomson to find out how the parts of an atom was arranged. Thomson on realised that the positively charged material in the atom and the negatively charged electrons would be attracted to each other, that the electron would combine to the positive material. Therefore he derived this to an analogy of a ‘‘plum pudding’’. He proposed that repulsion between the electrons would impose some ordered structure. If there was one electron it would be in the middle, two electrons would be on opposite sides and lager numbers would be located within rings.[5]
From this he discovered that no matter what metal the electrodes were made up of or what type of gas was in the tube, the mass to charge ratio was unchanged. He also deducted that there were two parts of atom held together by electrostatic attraction. A heavier part, responsible for the majority of mass of the atom and a negatively charged ‘corpuscle’ (electron).[4] This meant that Dalton’s model of an atom was incorrect, since the atom was not the smallest possible particle.
His experiment findings drove Thomson to find out how the parts of an atom was arranged. Thomson on realised that the positively charged material in the atom and the negatively charged electrons would be attracted to each other, that the electron would combine to the positive material. Therefore he derived this to an analogy of a ‘‘plum pudding’’. He proposed that repulsion between the electrons would impose some ordered structure. If there was one electron it would be in the middle, two electrons would be on opposite sides and lager numbers would be located within rings.[5]
Other Discoveries
Thompson was a great advocate of pure research, in contrast to applied research, declaring: ‘’Research in applied science leads to reforms, research in pure science leads to revolutions, and revolutions, whether political or industrial, are extremely profitable things if you are on the winning side’’. He also made the ground breaking discovery of the isotope. Which is one of two or more atoms having the same atomic number but differing in atomic weight. Thomson discovered Neon to be the first stable isotope, which helped explained basic aspects on radioactivity.[6]
Thompson was a great advocate of pure research, in contrast to applied research, declaring: ‘’Research in applied science leads to reforms, research in pure science leads to revolutions, and revolutions, whether political or industrial, are extremely profitable things if you are on the winning side’’. He also made the ground breaking discovery of the isotope. Which is one of two or more atoms having the same atomic number but differing in atomic weight. Thomson discovered Neon to be the first stable isotope, which helped explained basic aspects on radioactivity.[6]
[1] Atomicarchive.com. 2014. J.J. Thomson | Biography | atomicarchive.com. [online] Available at: http://www.atomicarchive.com/Bios/Thomson.shtml [Accessed: 10 Mar 2014].
[2] Sharwood, J. and Corrigan, D. 2007. Nelson chemistry. South Melbourne: Thomson Learning.
[3] Tuckerman, M. 2014. J.J. Thomson's experiment and the charge-to-mass ratio of the electron. [online] Available at: http://www.nyu.edu/classes/tuckerman/adv.chem/lectures/lecture_3/node1.html [Accessed: 10 Mar 2014].
[4] Sharwood, J. and Corrigan, D. 2007. Nelson chemistry. South Melbourne: Thomson Learning.
[5] CARL VILLANUEVA, J. 2009. Plum Pudding Model. [online] Available at: http://www.universetoday.com/38326/plum-pudding-model/ [Accessed: 10 Mar 2014].
[6] Encyclopaedia Britannica. 2013. Thomson atomic model. [online] Available at: http://www.britannica.com/EBchecked/topic/593128/Thomson-atomic-model [Accessed: 10 Mar 2014].
[2] Sharwood, J. and Corrigan, D. 2007. Nelson chemistry. South Melbourne: Thomson Learning.
[3] Tuckerman, M. 2014. J.J. Thomson's experiment and the charge-to-mass ratio of the electron. [online] Available at: http://www.nyu.edu/classes/tuckerman/adv.chem/lectures/lecture_3/node1.html [Accessed: 10 Mar 2014].
[4] Sharwood, J. and Corrigan, D. 2007. Nelson chemistry. South Melbourne: Thomson Learning.
[5] CARL VILLANUEVA, J. 2009. Plum Pudding Model. [online] Available at: http://www.universetoday.com/38326/plum-pudding-model/ [Accessed: 10 Mar 2014].
[6] Encyclopaedia Britannica. 2013. Thomson atomic model. [online] Available at: http://www.britannica.com/EBchecked/topic/593128/Thomson-atomic-model [Accessed: 10 Mar 2014].